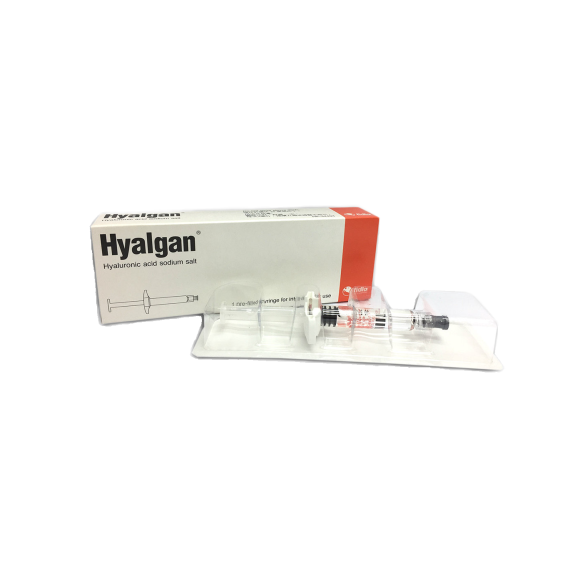
Hyalgan® contains hyaluronic acid, which is a natural polymer of glycosaminoglycan found in joint cartilage and synovial fluid. The hyaluronic acid in Hyalgan® is processed by a patented molecular filtration procedure. The efficacy and safety of Hyalgan® for different osteoarthritis (OA) joints, such as knee, ankle, shoulder, etc., have been well-proven in numerous clinical studies1.
In OA patients, the molecular weight and concentration of the hyaluronic acid in the synovial fluid are lower compared to healthy joint, reducing its protective effect2. Hyalgan® injection replenishes and boosts the normal production of synovial fluid in joints to relieve pain and other symptoms1.